
So, the reason why metals, such as aluminum, are coupled with oxides, such as ITO, are coupled is because they encourage a flow of electrons in a particular direction. This phenomenon is also explained using ionization energy if only a small amount of energy is necessary for an electron to be removed from a material, the material is said to have a low ionization energy. So, the electron donor and acceptor layers have a low and high electron affinity respectively. Non-metals are known to have a high electron affinity for this reason. 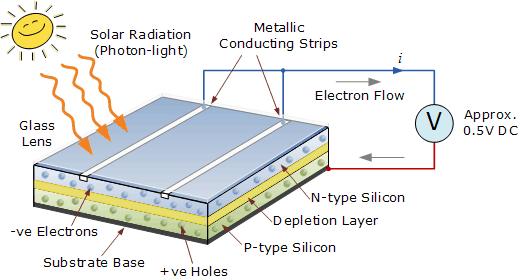
Other materials, such as non-metals, have their electrons much more tightly bound to the nucleus, meaning that they preferably gain electrons. Metals have loosely bound valence electrons, which makes it probable for them to lose an electron rather than gaining one, giving metals a low electron affinity. The advantage of this style is that each of the two organic layers have a different electron affinity and ionization energy, meaning that additional force is generated at the donor-acceptor interface. The two layers are classified as the electron donor and acceptor layer, which is shown in Figure 2. Because of this phenomenon, not many of the electrons participate in the overall current needed to power a source.ĭual layer OSCs are a significant upgrade to single layer, as an additional polymer layer is added between the electrodes. Even when exciton bonds are broken, the energy to do so is typically just barely enough, which most often results in the electron and hole recombining. Unfortunately, single layer OSCs are highly inefficient, as the potential difference across the polymer is not strong enough to thoroughly break a significant amount of the excitons. The potential difference created by the electrodes aids the separation process of the excitions by breaking the electrostatic bonds, allowing electrons to migrate to the cathode and holes to migrate the the anode. Once photons become absorbed in the polymer layer, excitons are formed by promoting electrons into the LUMO. Some types of materials used for the single polymer layer include phthalocyanine, polyflourenes, polypyrenes, and polythiophenes. The most basic type of OSC is the single layer, as it is composed of a single polymer layer sandwiched in between two electrodes. Single Layer OSC Figure 2: Single Layer OSC Diagram In order to truly pursue the green movement, OSCs must be heavily implemented and researched, as they are the most environmentally and human friendly alternate energy source available today. 
Some significant advantages of OSCs are that they do not contain toxic heavy metals, making them easy to process and environmentally friendly, they are quite flexible, and they are very low in processing and material costs. OSCs, while still not as efficient as ISCs, will continue to become more efficient as they are more heavily researched. The band gap energy of organic materials used in OSC is usually found to be within the 1-4eV range. Since there is an energy difference between the HOMO and LUMO, this creates what is sometimes known as an organic band gap. For practical purposes, the HOMO and LUMO can be compared to the valence and conduction band of an ISCs respectively. Instead, they function using the highest occupied molecular orbital(HOMO) and lowest unoccupied energy orbital(LUMO). While ISCs utilize the band gap of semiconductors, polymers in OSCs do not have band gaps. If too many particles, such as pollen or dust, accumulate on the surface of the solar cell, photons scatter instead of being absorbed, giving way to a much lower absorption. Even on a clear, sunny day with minimal pollution in the air, solar cell surfaces must be cleaned in order to remove particulates to obtain maximum photon absorption. It is important to realize that many of the photons emitted from the sun will not be absorbed in the solar cell, as photons can be absorbed by particles or molecules in the air, such as dust, oxygen, water, methane, or carbon dioxide. \) will be about 0.5 to 0.6 volts, thus we could expect to get maybe around 2.\] where h is Planck's constant, ν is the frequency of the photon, c is the speed of light, and λ is the wavelength of the photon.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
